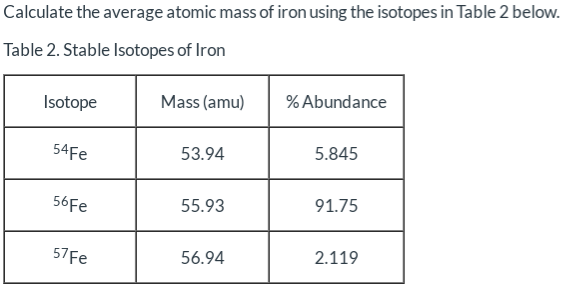
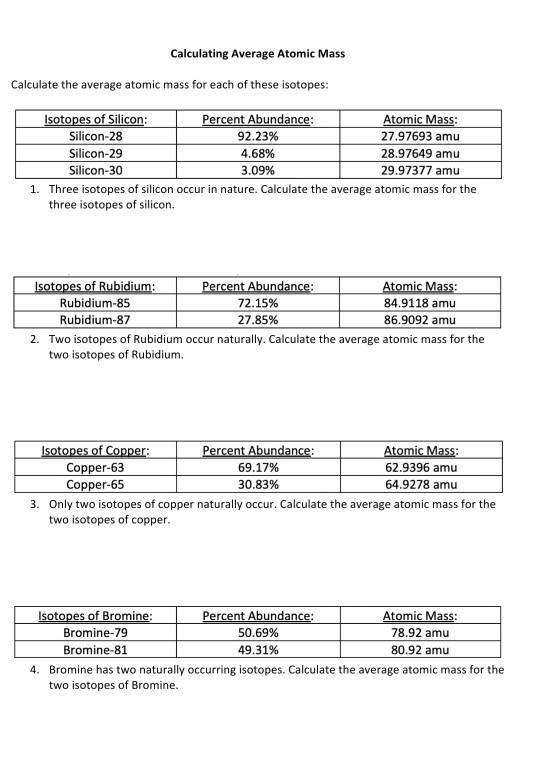
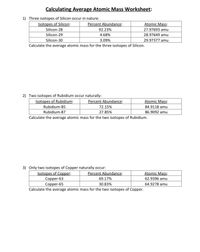
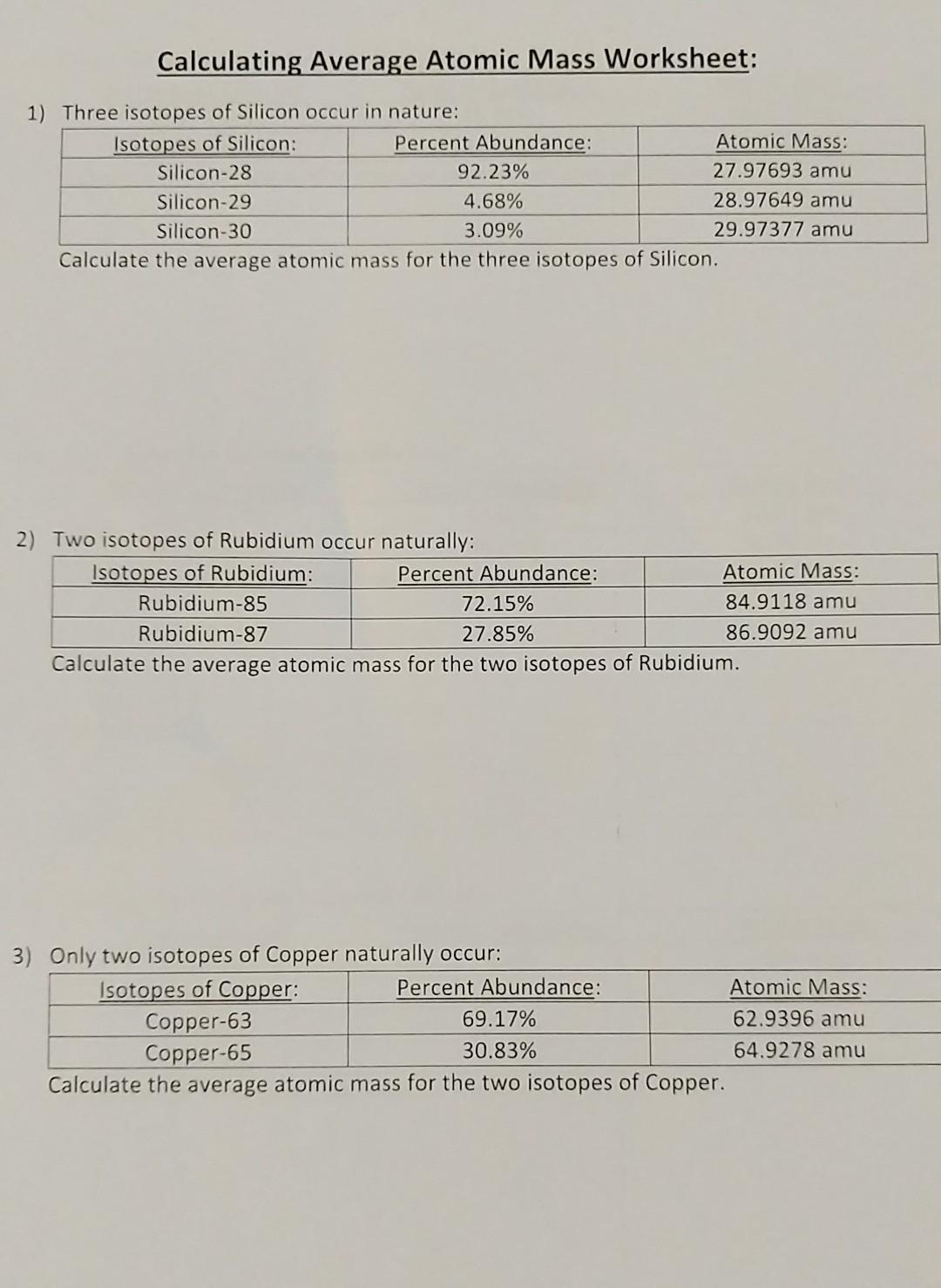
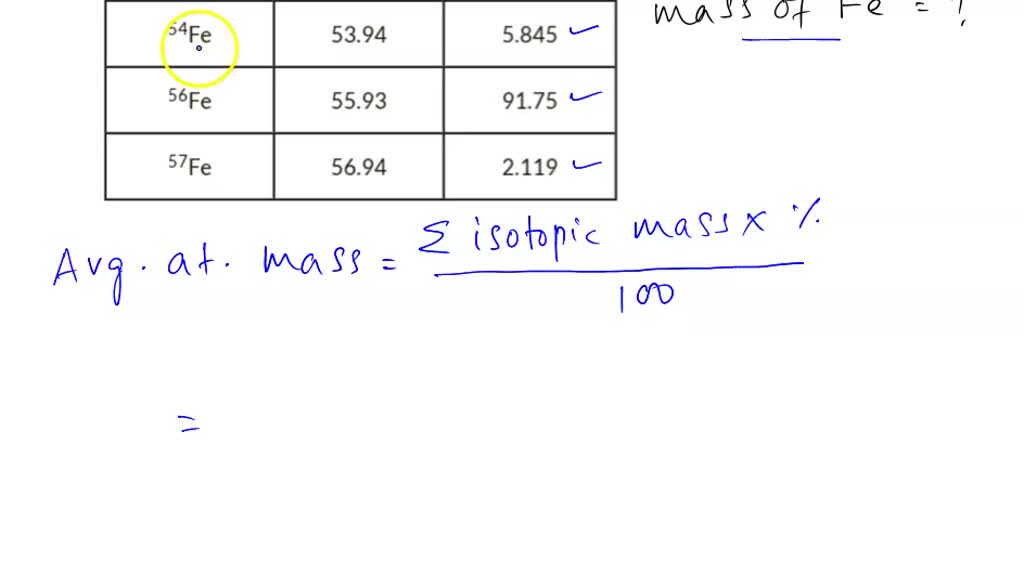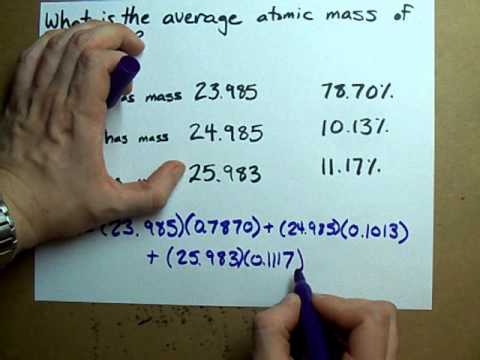_Copy of Average Atomic Mass.docx - chem.docx - Name: _ Simulation: Isotopes & Calculating Average Atomic Mass Background In this investigation you will | Course Hero

simulation_isotopes_average_atomic_mass_student (1).docx - Name: _ Simulation: Isotopes & Calculating Average Atomic Mass Background In this | Course Hero
Why while calculating the average atomic mass of chlorine didn't we divide it by 2 as in the average denominator has a sum of the total outcomes? - Quora

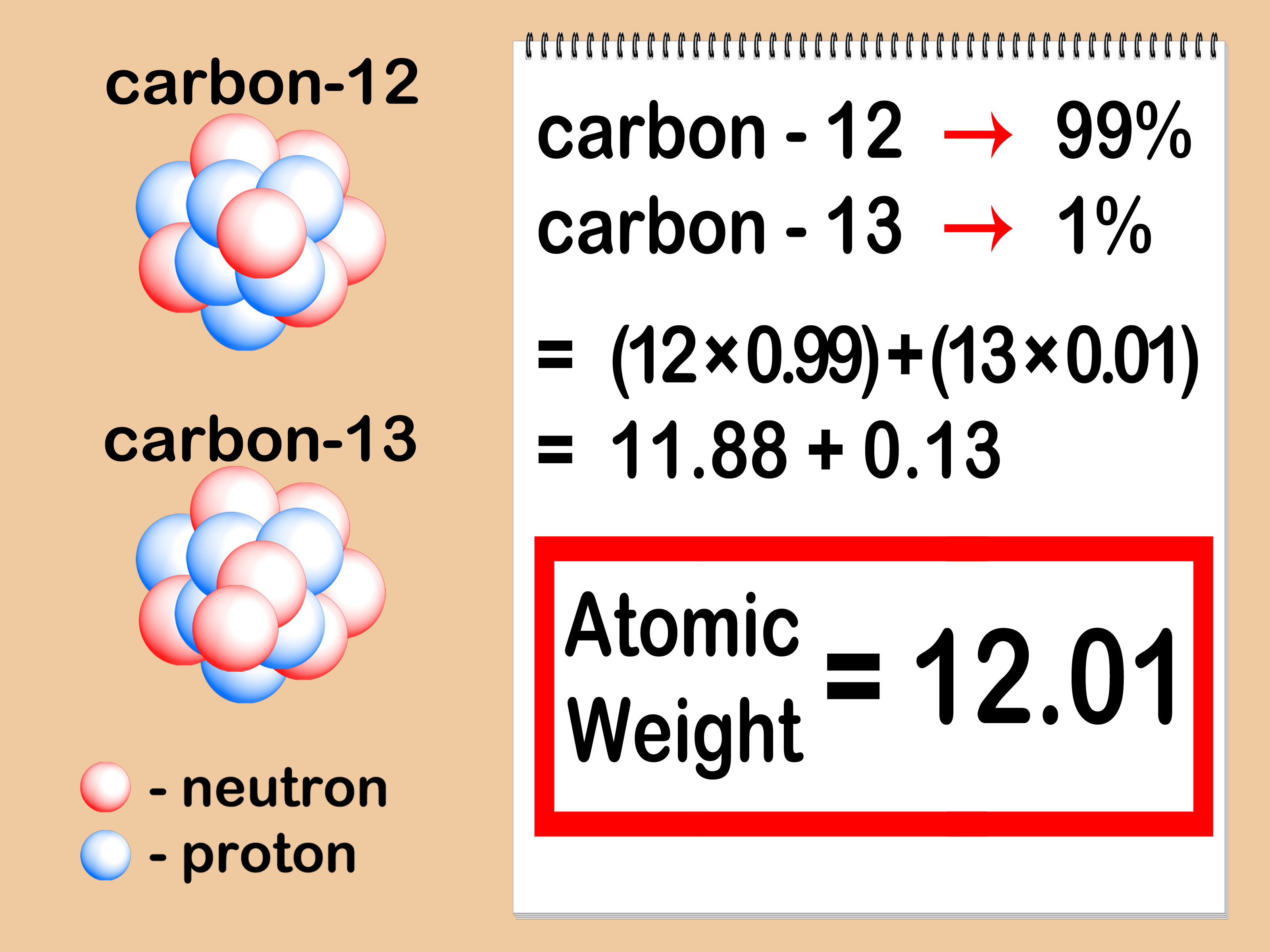
Average Atomic Mass In nature, most elements are a mixture of different isotopes The mass of a sample of an element is a weighted average of all the isotopes. - ppt download